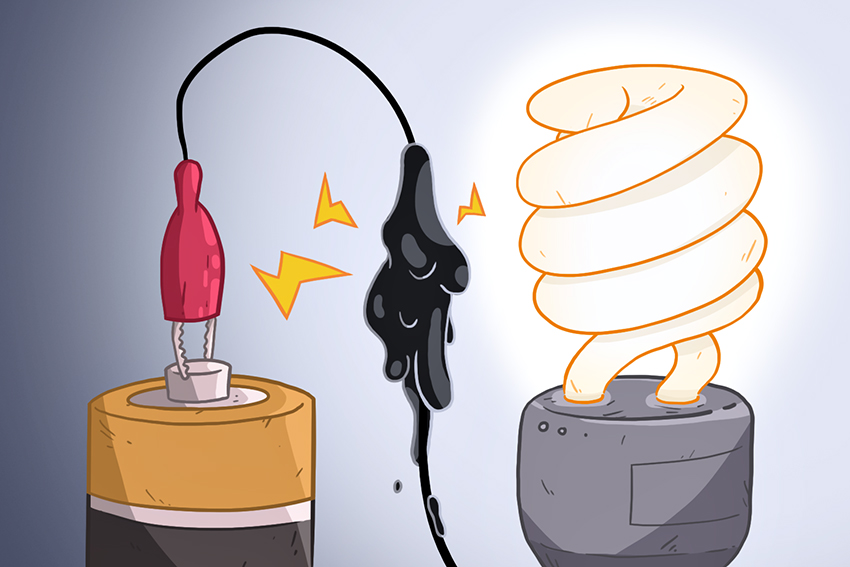
The future of electronics depends on the capabilities of next generation batteries.
The materials of these batteries typically expand during charging, which eventually damages battery electrodes. UT engineers have created a self-healing gel that repairs broken circuits and conducts electricity.
Guihua Yu, assistant mechanical engineering professor, contributed to the solution of this battery problem.
“Scientists and engineers really want to mimic what human skin can do,” Yu said. “We know that with small wounds take a couple days to heal, but it is a natural process.”
This gel has potential use in flexible electronics and complex next generation batteries, according to the paper published in Nano Letters.
This gel contains two parts: a conducting polymer gel and a supramolecular gel, a group of bonded molecules that provides the self-healing properties. Yu said that the combination of these materials makes this gel unique.
“Most of the conventional self-healing gels don’t have good electronic conducting properties,” Yu said. “This limits their applications in electronics.”
Yu’s team worked with a special class of polymer, called a conjugated polymer, which is adept at conducting electricity. This polymer then combines with the supramolecular gel to form the hybrid substance. The supramolecular gel contains zinc ions and terpryidine, which together eliminate the need for external pressure or heat for the gel to heal.
The next step after synthesizing this supramolecular gel was binding it to the conductive polymer. Ye Shi, a material science postdoctoral student and member of the Yu lab team, said that this took time to accomplish.
“Our material is a hybrid containing two parts,” Shi said. “So the difficulty was finding the perfect combination of elements, adjusting the ratio between the two components and finding good methods of synthesizing and combining them.”
The current product takes just minutes to repair itself. The gel is applied to circuit junction points, places in the battery that often break. When damage occurs, the supramolecular components of the gel allow it to reform and continue the flow of electricity, without any changes to the mechanical properties. Yu says this gel has applications in many different electronics.
“Today the base material for electronics is silicon, but we know from basic chemistry that silicon is a semi-conductor and is brittle,” Yu said. “Often when using silicon for flexible electronics, the electronic parts will break. Here is where our self-healing gel is very helpful.”
More advanced inventions such as wearable electronics and robots can also benefit from this gel.
“Robots have lots of different electrical wiring inside and if anything breaks, very extensive work is required to fix it,” Yu said. “We want these robots to do things in a human manner and not having to manually fix every broken element can achieve this and help such devices last longer.”
The team published their work in multiple papers last fall, but continue working on more efficient ways to synthesize the gel. Yu said that the process currently takes a few weeks and is not scalable. He is uncertain about the cost and implementation into consumer markets.
“It just depends on how the gel is synthesized and how efficiently it can be processed,” Yu said.
Despite his reservations, Yu said that the gel will have a serious impact in the field of electronics.
“I see great potential in many areas,” Yu said. “Because this gel is conductive, it is relatively straightforward to apply to flexible or wearable electronics. This gel allows parts to heal, which in turn makes your devices last longer and more cost effective.”