Editor's Note: This story first appeared in The Daily Texan's February 2 print edition.
Researchers at the University of Texas at Austin published a study Jan. 28 explaining how remdesivir, the only COVID-19 antiviral drug approved by the Food and Drug Administration, works to fight COVID-19 infection.
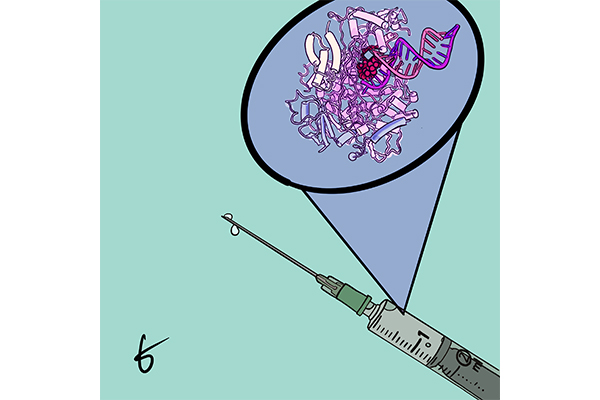
The COVID-19 virus is able to infect a person by copying itself with other cells in the body, said co-author Kenneth Johnson, professor of molecular biosciences. COVID-19 has an error-correction mechanism that allows it to remove mistakes that would stop the virus from spreading, Johnson said. This finding could potentially allow drug companies to develop a drug that is easier to take to treat COVID-19. Currently, the drug requires an injection.
Remdesivir is an antiviral drug that was first worked on and approved for the Ebola virus, said David Taylor, assistant professor of molecular biosciences.
The study sheds light on remdesivir and how it combats the COVID-19. Johnson said remdesivir blocks COVID-19’s RNA polymerase, which would have allowed the virus to copy itself onto other cells. Despite COVID-19’s error-correction mechanism, the virus isn’t able to remove remdesivir because it is rapidly incorporated and buried by other molecules.
Taylor said he compares the reaction between COVID-19’s polymerase and remdesivir to a photocopier and a paper jam.
“The polymerase is like a photocopier,” Taylor said. “It wants to keep making copies of the viral genome so it can keep replicating. Remdesivir causes one of those papers to get jammed in the machine, so you can’t make more copies and can’t keep relocating and infecting.”
Biochemistry graduate student Tyler Dangerfield said the team was able to identify remdesivir’s trick by recreating the process of a patient infected with COVID-19 receiving remdesivir in a lab dish. The team froze the reaction between the polymerase and the remdesivir, allowing them to observe how remdesivir does its job.
Dangerfield, who worked to prepare the COVID-19 polymerase, said this was the most challenging part of the project for him, taking him three months to complete.
“A lot of the times when you try to study proteins that are made in bacteria, they are not made that well,” Dangerfield said. “That was really kind of (a) frustrating time because you don’t really know if it’s going to work or not, you just have to try.”
Taylor said as more COVID-19 strain mutations emerge, it’s only a matter of time until researchers need to develop more effective antiviral treatments against the virus. The discovery of remdesivir’s process will allow drug companies to find ways to make more effective antiviral drugs.
Johnson said if patients with COVID-19 realize they are infected early on, they can potentially go to a hospital and get treated with doses of remdesivir through injection for a four to five day period.
“It would be much nicer if you got a positive COVID test and they could give you a pill to take home with you, and if you start feeling (the) earliest symptoms, you just take the pill,” Johnson said.